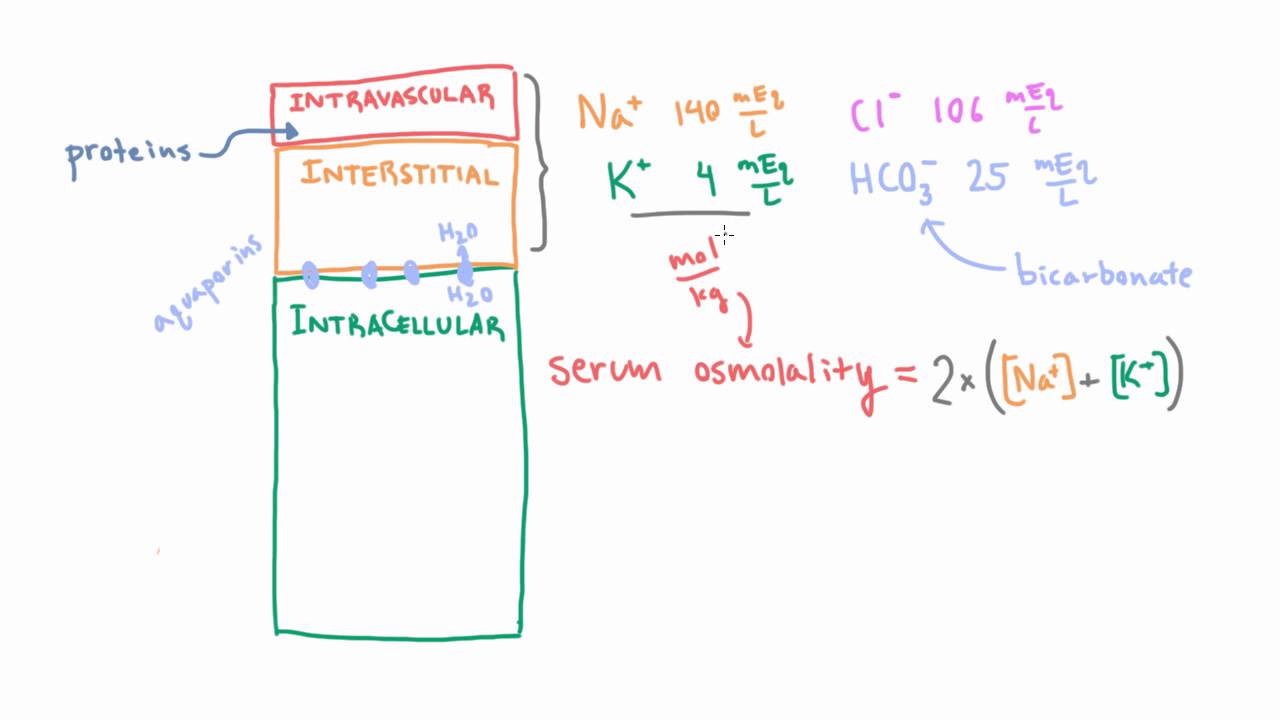
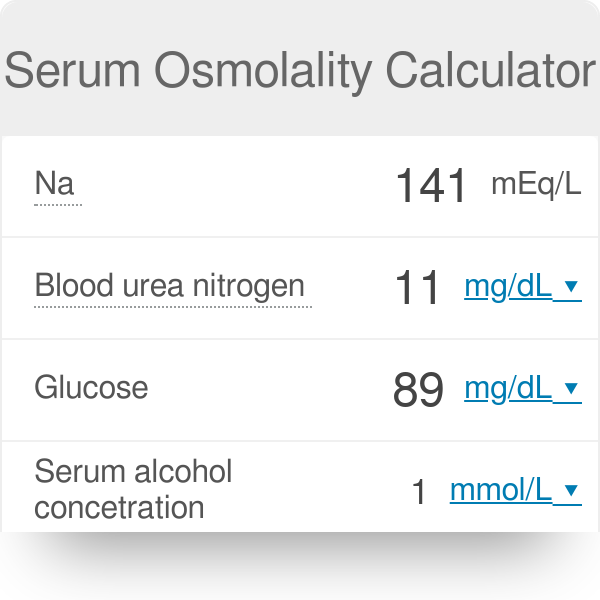
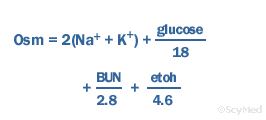SOLVED: Example Calculation of Plasma Osmolality Estimate from the following data: sodium, 135 mEq/L; blood urea nitrogen, 14 mg/dL; and glucose, 90 mg/dL. Osmolality = (2 x sodium mEq/L) + (blood urea
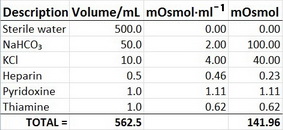
![PDF] Comparison of methods for calculating serum osmolality form chemical concentrations, and the prognostic value of such calculations. | Semantic Scholar PDF] Comparison of methods for calculating serum osmolality form chemical concentrations, and the prognostic value of such calculations. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6e2d1656fc822211705e560511c8cb3abdebb75e/2-Table1-1.png)
PDF] Comparison of methods for calculating serum osmolality form chemical concentrations, and the prognostic value of such calculations. | Semantic Scholar
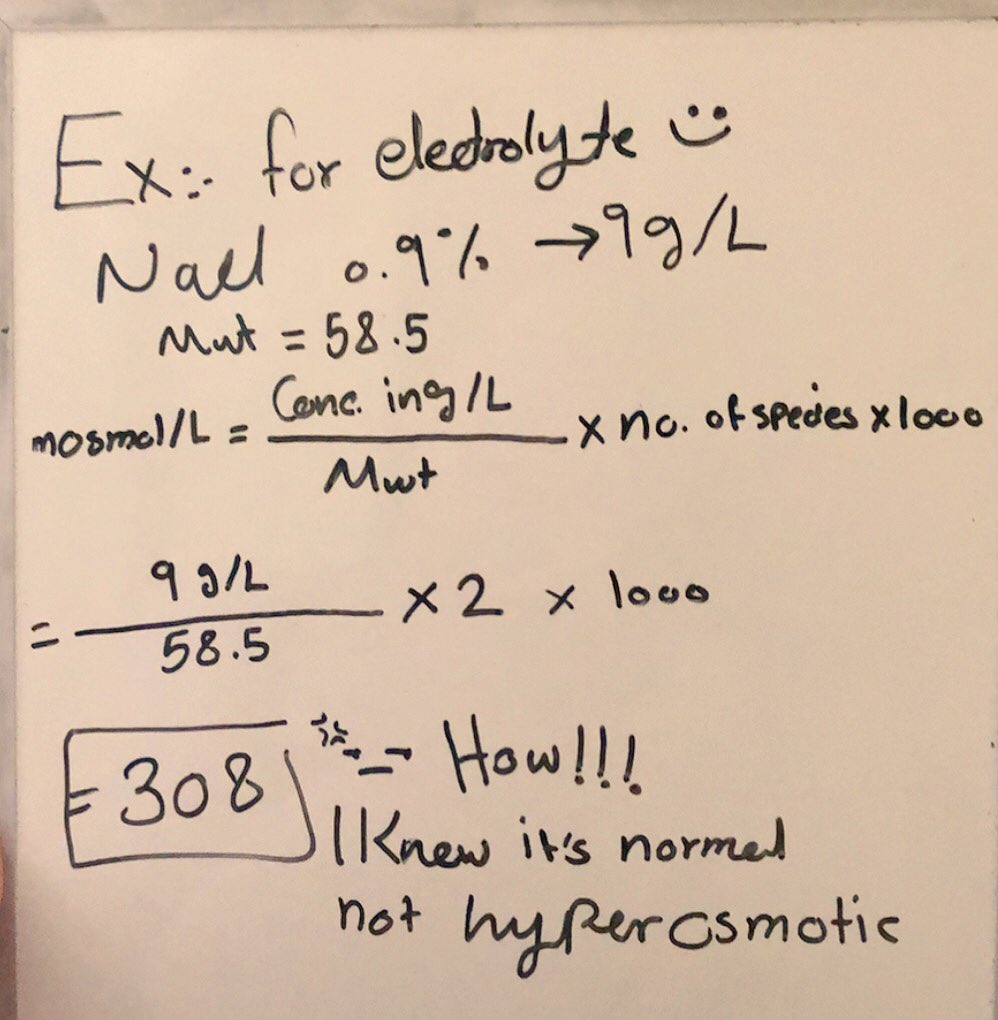
ph.jawzaa|ص.جوزاء on Twitter: "Example 1: Calculate the ideal osmolarity of 0.9 % NaCl injection? Because of bonding forces, however, n is slightly less than 2 for solutions of sodium chloride at this
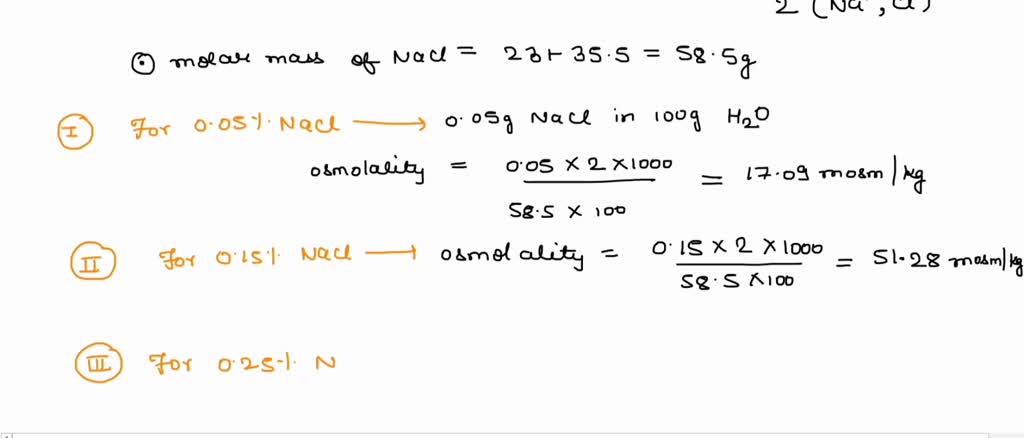
SOLVED: Calculate the osmolality for each of these six solutions: 0.05%, 0.15%, 0.25%, 0.6%, 0.9%, 1.2% NaCl. Which concentration(s) is/are closest to that of plasma (290 mOsm/kg)?