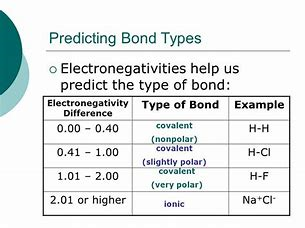
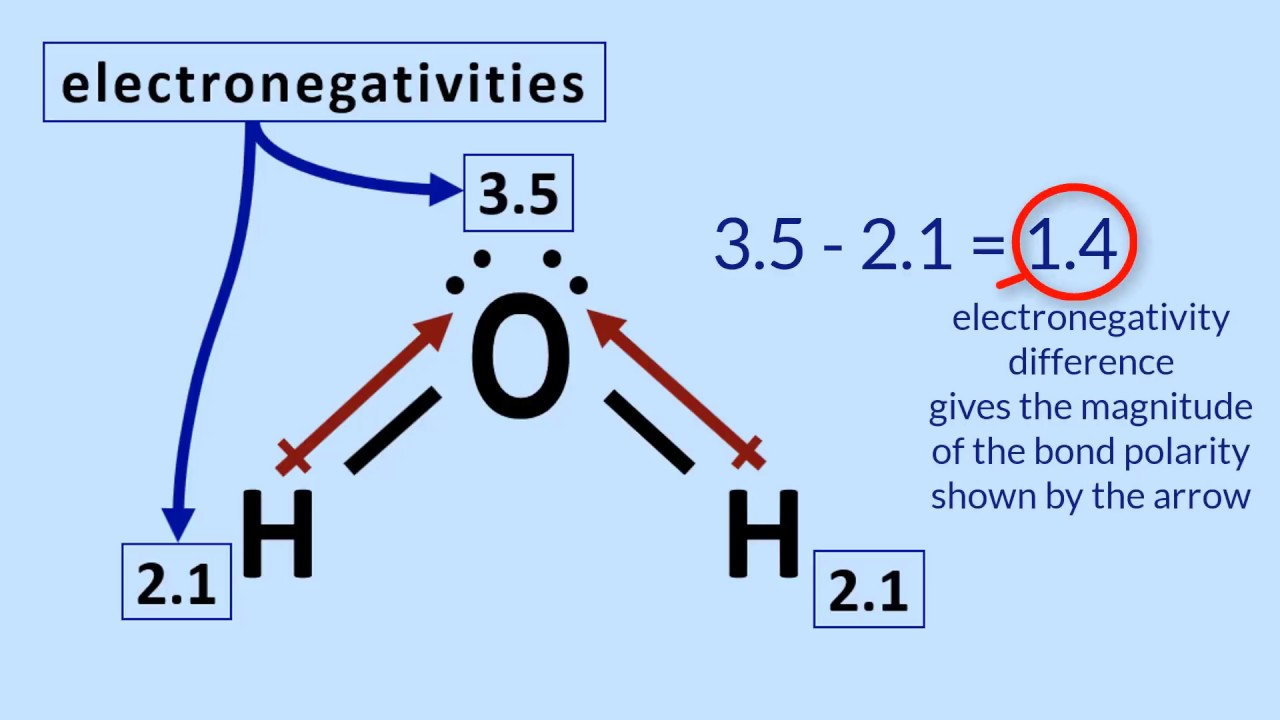
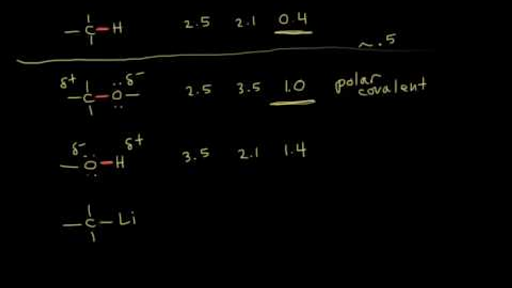
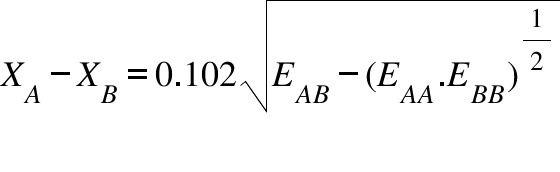Welcome to Chem Zipper.com......: The electronegativity of F and H are 4.0 and 2.1 respectively. The percentage ionic character in H and F bond is.

Calculate the electronegativity (10 time) of fluorine on Pauling scale form the following data: EH - H = 104.2kcal mol^-1 EF - F = 36.6kcal mol^-1 EH - F = 134.6kcal mol^-1 Given = XH = 2.1

Calculate the electronegativity value of chlorine on Mulliken's scale, given that `IP = 13.0 eV` and - YouTube

SOLVED: Calculate the electronegativity difference of the following compounds below and arrange them in an increasing bond strength. a HCI b NaBr C. Cl2 d. HzO e MgO