
Calculate the entropy change in surroundings when 1.00 mol of H2O (l) is formed under standard conditions at 298k, Given ,H^o = - 286kJ mol^-1

Oxygen gas weighing 64 g is expanded from 1 atm to 0.25 atm at 30^(@)C calculate entropy change, assuming the gas to be ideal.

How to Calculate the Entropy Change for a Chemical or Physical Process Based on Absolute Entropies | Chemistry | Study.com

Calculate the entropy change in surroundings when 1.00 mol of H2O (l) is formed under standard.... - YouTube

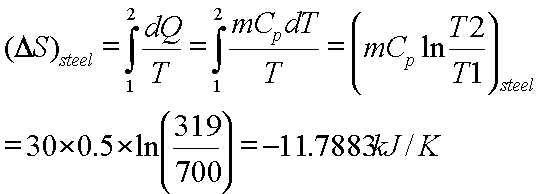



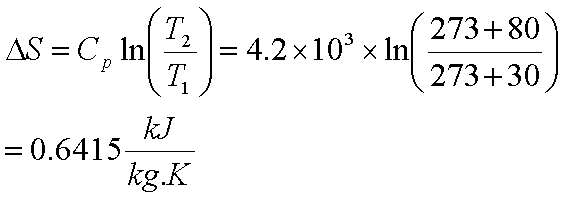





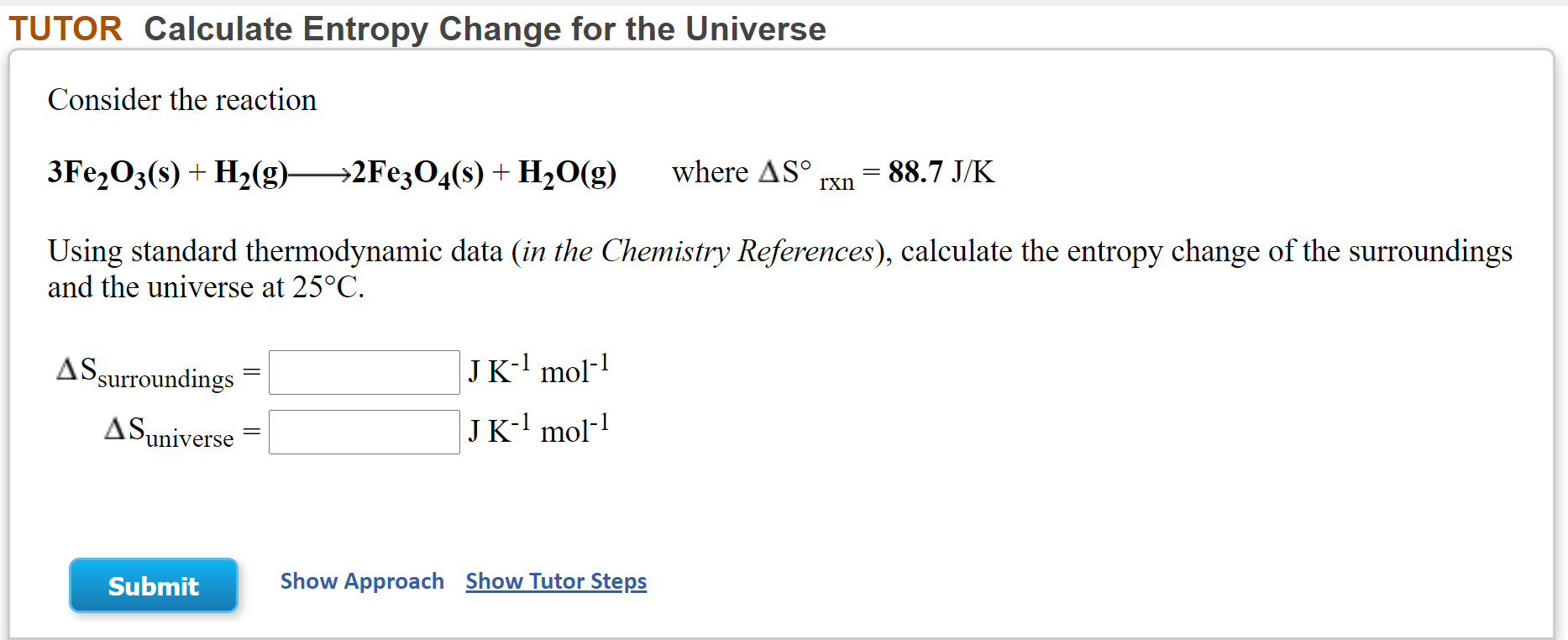
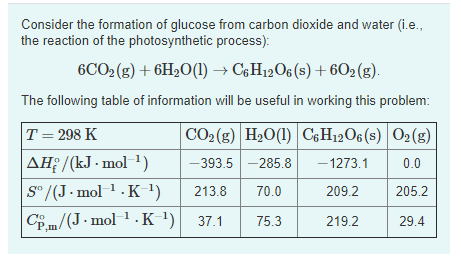


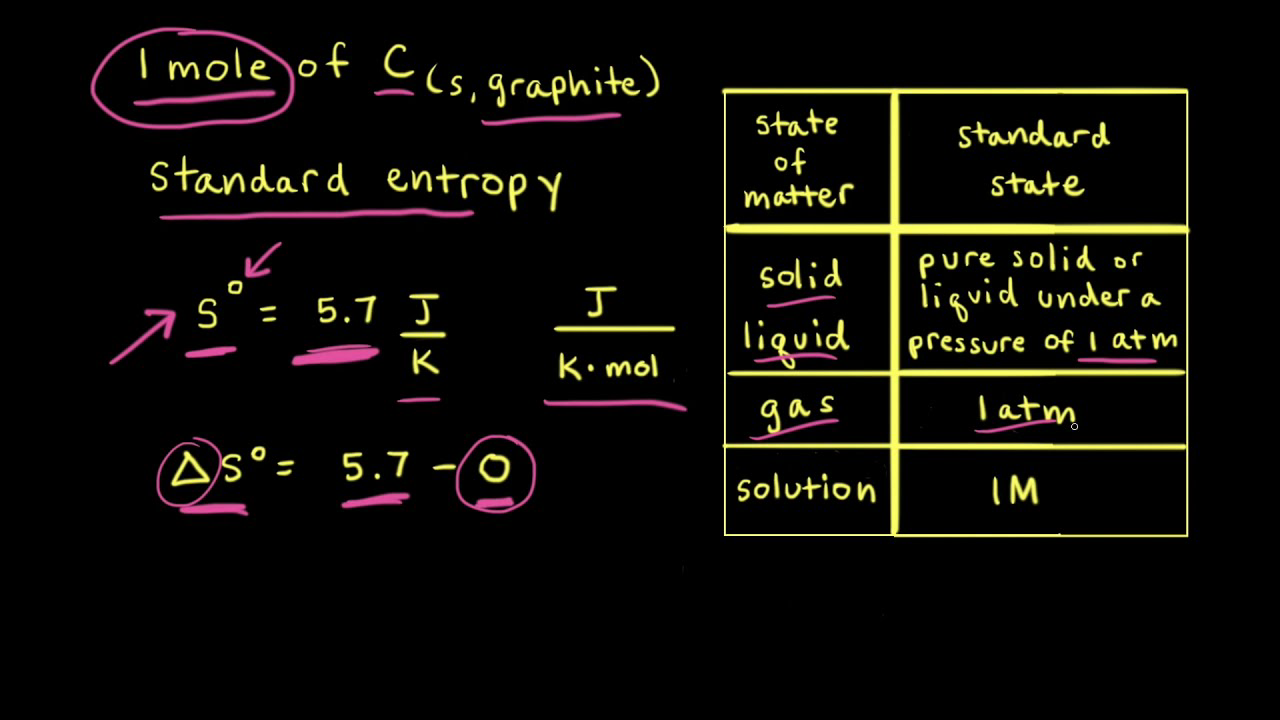




![15.2/R1.4 Calculate the standard entropy change for a reaction [HL IB Chemistry] - YouTube 15.2/R1.4 Calculate the standard entropy change for a reaction [HL IB Chemistry] - YouTube](https://i.ytimg.com/vi/IwRy4iYVQLI/maxresdefault.jpg)