SOLVED: 'Calculate the amount of heat required to heat 357 grams of ethylene glycol (antifreeze) from 0 to 110 The specific heat of ethylene glycol is 2.42 Jg Select one: a. 71.5
![An antifreeze solution is prepared from `222.6 g` of ethylene glycol `[C_(2)H_(4)(OH)_(2)]` and ... - YouTube An antifreeze solution is prepared from `222.6 g` of ethylene glycol `[C_(2)H_(4)(OH)_(2)]` and ... - YouTube](https://i.ytimg.com/vi/qDRYyKBs2_U/maxresdefault.jpg)
An antifreeze solution is prepared from `222.6 g` of ethylene glycol `[C_(2)H_(4)(OH)_(2)]` and ... - YouTube
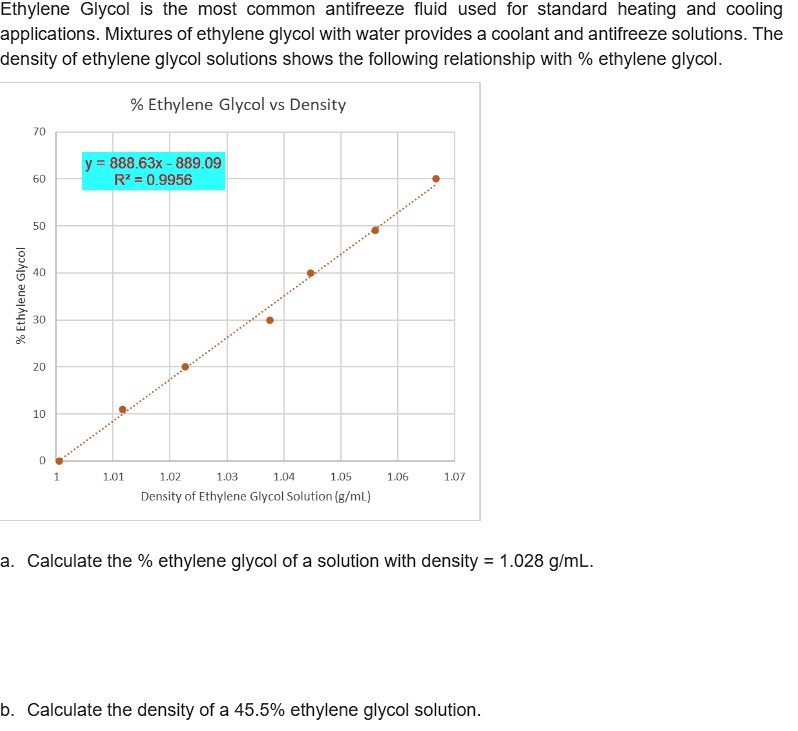
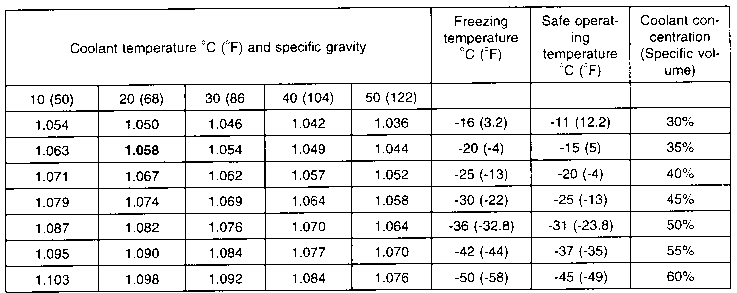
SOLVED: Ethylene Glycol is the most common antifreeze fluid used for standard heating and cooling applications. Mixtures of ethylene glycol with water provides a coolant and antifreeze solutions. The density of ethylene

Ethylene glycol (molar mass = 62gmol^-1) is a common automobile antifreeze. Calculate the freezing point of a solution containing 12.4 g of this substance in 100 g of water. Would it be

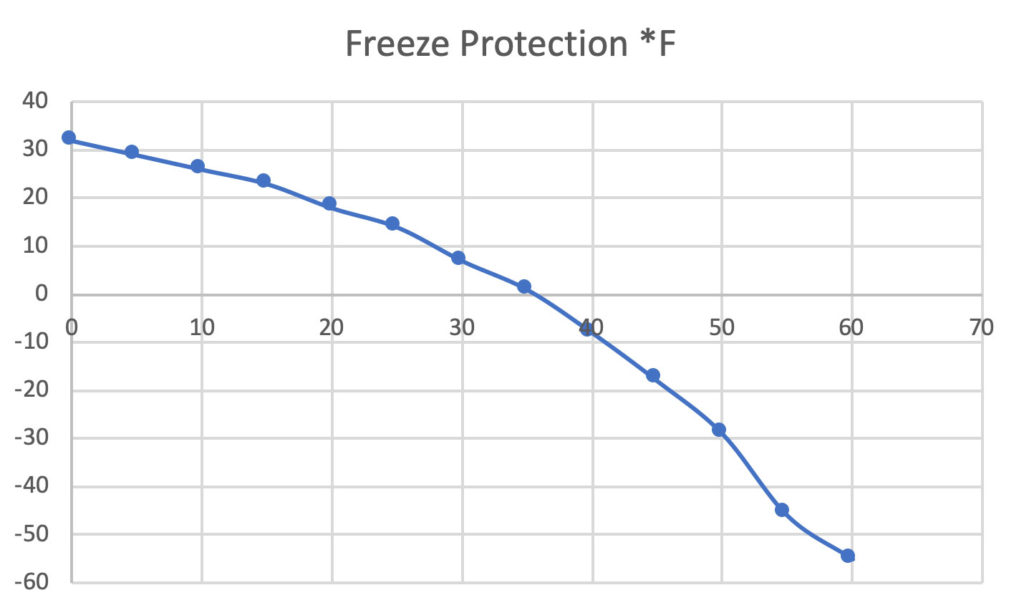
Ethylene glycol is used as an antifreeze agent. Calculate the amount of ethylene glycol to be added to 4 kg of water to prevent it from freezing at - 6^∘ C.(Kf for